|
Sometimes aqueous is never used and the question writer simply assumes you know everything is happening in aqueous solution. The word 'aqueous' could be used as well as 'precipitate.' Sometimes aqueous is used for the reactants, but assumed for the products. The wording in problems like the above can vary somewhat. Here are two examples:īarium chloride solution reacts with sodium sulfate solution to make solid barium sulfate and aqueous sodium chlorideĪqueous solutions of hydrochloric acid and sodium hydroxide react to produce sodium chloride and water 
Many times, problems of this type will start out with an equation in words. This type of equation shows the full formula for each substance involved (or the full name of each substance), without reference to a substance being ionic or molecular. Or, perhaps, a future lab partner who learned it one way, while you had learned it a different way. You have to know them all (even the one-offs) because you never know what a particular teacher/textbook might use. They are (a) 'empirical equation' and (b) 'non-ionic equation.' In my years of doing chemistry stuff, I have seen two one-off names for what I call the complete molecular equation. 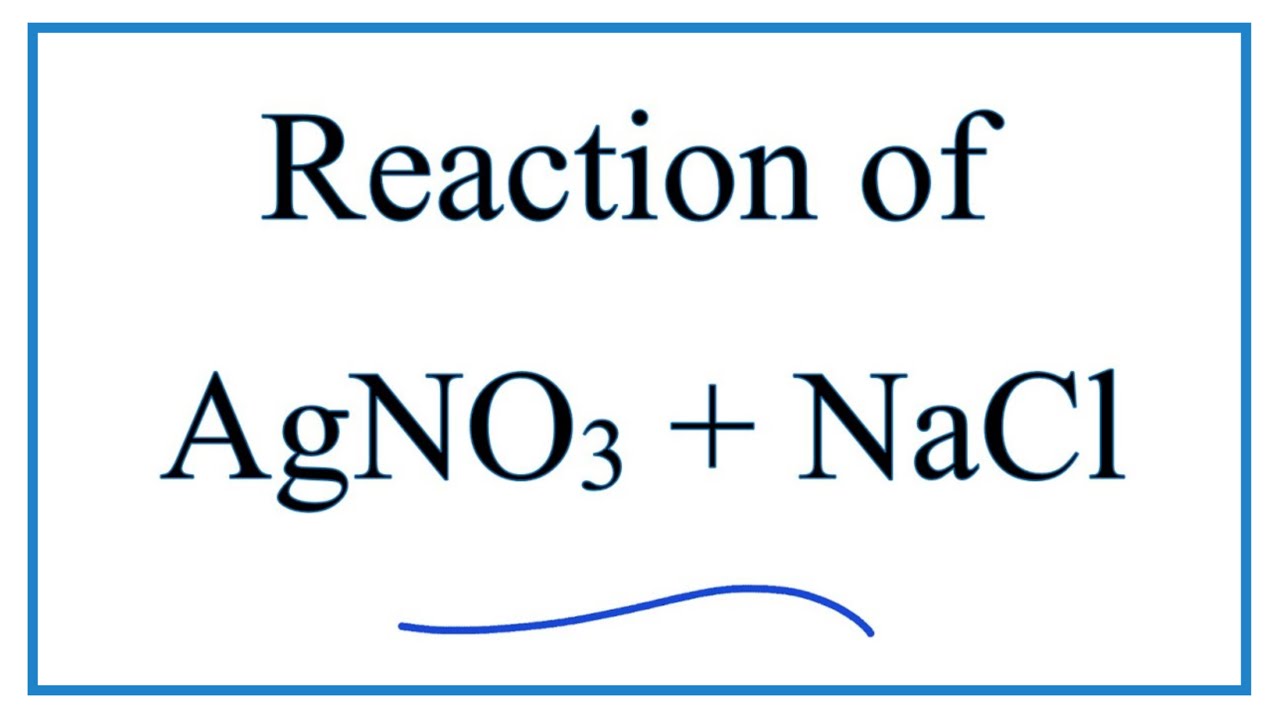
These equations can also be called 'complete formula equations,' 'total formula equations,' or simply 'formula equations.' There is no standard name. If you go over there, make sure you ignore the late-1990's style and concentrate on the information. It has a lot of good information, said in a different sequence than I do below. How to Write Ionic Equations is an extensive discussion of the topic. 
ChemTeam: Equations: Complete Molecular, Complete Ionic and Net Ionic Equations: Complete Molecular, Complete Ionic and Net Ionic Ten Problems Fifteen Problems Twenty-Five Problems All NR Equations & Reaction Types menu
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
